概要
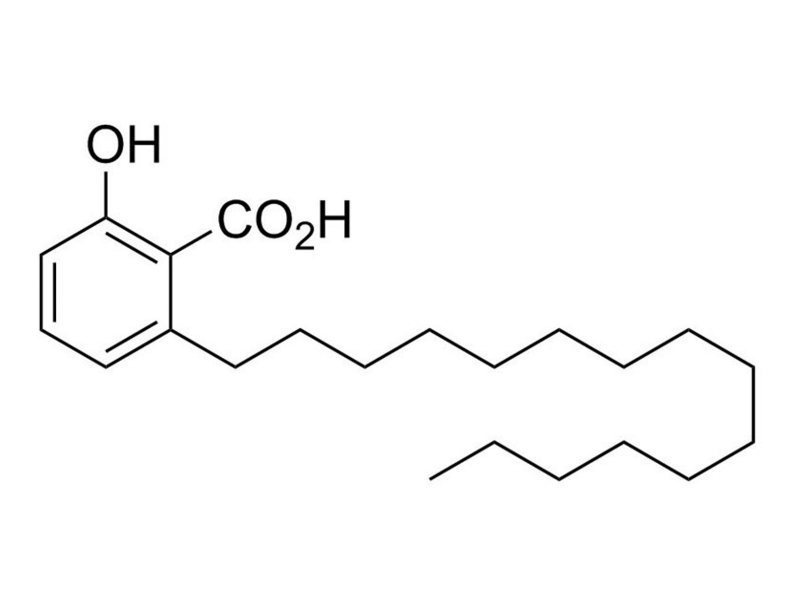
Anacardic Acid is a 6-alkyl salicyclic acid that inhibits the histone acetyltransferase (HAT) activity of the transcription co-activators p300 and p300/CREB-binding protein-associated factor (pCAF; IC₅₀ values of 8.5 and 5 µM, respectively; Balasubramanyam et al.). Anacardic Acid inhibits HAT-dependent transcription and protein SUMOylation (Cui et al.; Fukuda et al.). In addition, Anacardic Acid is an activator of Aurora kinase A-mediated phosphorylation of Histone H3 (Kishore et al.). At higher concentrations (25 µM), Anacardic Acid suppresses NF-κB activation and inhibits IκB-α phosphorylation (Sung et al.).
DIFFERENTIATION
· Enhances cardiomyocyte differentiation from mouse embryonic stem cells (Re et al.).
IMMUNOLOGY
· Induces macrophage activation (Gnanaprakasam et al.).
· Exhibits inhibitory and bactericidal activities against methicillin-resistant Staphylococcus aureus (Muroi & Kubo).
CANCER RESEARCH
· Potentiates the apoptosis induced by cytokine and chemotherapeutic agents in cancer cells (Sung et al.).
· Sensitizes tumor cells to ionizing radiation in vitro (Sun et al).
DIFFERENTIATION
· Enhances cardiomyocyte differentiation from mouse embryonic stem cells (Re et al.).
IMMUNOLOGY
· Induces macrophage activation (Gnanaprakasam et al.).
· Exhibits inhibitory and bactericidal activities against methicillin-resistant Staphylococcus aureus (Muroi & Kubo).
CANCER RESEARCH
· Potentiates the apoptosis induced by cytokine and chemotherapeutic agents in cancer cells (Sung et al.).
· Sensitizes tumor cells to ionizing radiation in vitro (Sun et al).
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | Anacardic Acid | 73192, 73194 | All | English |
| Safety Data Sheet | Anacardic Acid | 73192, 73194 | All | English |
数据及文献
Publications (9)
Endocrine 2015 NOV
Anacardic acid and thyroid hormone enhance cardiomyocytes production from undifferentiated mouse ES cells along functionally distinct pathways.
Abstract
Abstract
The epigenetics of early commitment to embryonal cardiomyocyte is poorly understood. In this work, we compared the effect of thyroid hormone and that of anacardic acid, a naturally occurring histone acetylase inhibitor, or both in combination, on mouse embryonic stem cells (mES) differentiating into embryonal cardiomyocyte by embryoid bodies (EBs) formation. Although the results indicated that anacardic acid (AA) and thyroid hormone were both efficient in promoting cardiomyocyte differentiation, we noticed that a transient exposure of mES to AA alone was sufficient to enlarge the beating areas of EBs compared to those of untreated controls. This effect was associated with changes in the chromatin structure at the promoters of specific cardiomyogenic genes. Among them, a rapid induction of the transcription factor Castor 1 (CASZ1), important for cardiomyocytes differentiation and maturation during embryonic development, was observed in the presence of AA. In contrast, thyroid hormone (T 3) was more effective in stimulating spontaneous firing, thus suggesting a role in the production of a population of cardiomyocyte with pacemaker properties. In conclusion, AA and thyroid hormone both enhanced cardiomyocyte formation along in apparently distinct pathways.
International immunopharmacology 2015 DEC
The anacardic 6-pentadecyl salicylic acid induces macrophage activation via the phosphorylation of ERK1/2, JNK, P38 kinases and NF-κB.
Abstract
Abstract
Amphipterygium adstringens is a plant traditionally used to treat gingivitis, gastric ulcer and even gastric cancer but the mechanism involved in the regulation of the immune response is not elucidated yet. The 6-pentadecylsalicylic acid (6SA) is the main anacardic acid found in A. adstringens. In order to evaluate the immune-modulatory abilities of 6SA, we used mouse splenocytes and determined the phosphorylation of the transcription factor NF-κB and MAP kinases ERK1/2, JNK and p38 in helper and cytotoxic T cells, natural killer (NK) cells and F4/80(+) macrophages. Treatment with 6SA was not cytotoxic as measured by both trypan blue exclusion and tetrazolium salts (MTT) tests. Additionally, 6SA did not alter the proportion of helper and cytotoxic T lymphocytes, NK cells or macrophages. Moreover, 6SA treatment significantly increased the phosphorylation of ERK1/2, JNK, P38 and NF-κB mainly in macrophages. In this cells (peritoneal macrophages), treatment with 6SA increased the secretion of nitric oxide (NO), interleukin (IL)-6 and tumour necrosis factor (TNF)-α and decreased the secretion of IL-4 and IL-10 depending on MAPK and NF-κB phosphorylation. In addition, 6SA increased the migration and phagocytic activity of macrophages also depending on the phosphorylation of different kinases. These data suggest that 6SA induces the classical activation pathway in macrophages via the phosphorylation of MAP kinases and NF-κB thus activating the adaptive immune system.
Chemistry & biology 2009 FEB
Ginkgolic acid inhibits protein SUMOylation by blocking formation of the E1-SUMO intermediate.
Abstract
Abstract
Protein modification by small ubiquitin-related modifier proteins (SUMOs) controls diverse cellular functions. Dysregulation of SUMOylation or deSUMOylation processes has been implicated in the development of cancer and neurodegenerative diseases. However, no small-molecule inhibiting protein SUMOylation has been reported so far. Here, we report inhibition of SUMOylation by ginkgolic acid and its analog, anacardic acid. Ginkgolic acid and anacardic acid inhibit protein SUMOylation both in vitro and in vivo without affecting in vivo ubiquitination. Binding assays with a fluorescently labeled probe showed that ginkgolic acid directly binds E1 and inhibits the formation of the E1-SUMO intermediate. These studies will provide not only a useful tool for investigating the roles of SUMO conjugations in a variety of pathways in cells, but also a basis for the development of drugs targeted against diseases involving aberrant SUMOylation.
Blood 2008 MAY
Anacardic acid (6-nonadecyl salicylic acid), an inhibitor of histone acetyltransferase, suppresses expression of nuclear factor-kappaB-regulated gene products involved in cell survival, proliferation, invasion, and inflammation through inhibition of the i
Abstract
Abstract
Anacardic acid (6-pentadecylsalicylic acid) is derived from traditional medicinal plants, such as cashew nuts, and has been linked to anticancer, anti-inflammatory, and radiosensitization activities through a mechanism that is not yet fully understood. Because of the role of nuclear factor-kappaB (NF-kappaB) activation in these cellular responses, we postulated that anacardic acid might interfere with this pathway. We found that this salicylic acid potentiated the apoptosis induced by cytokine and chemotherapeutic agents, which correlated with the down-regulation of various gene products that mediate proliferation (cyclin D1 and cyclooxygenase-2), survival (Bcl-2, Bcl-xL, cFLIP, cIAP-1, and survivin), invasion (matrix metalloproteinase-9 and intercellular adhesion molecule-1), and angiogenesis (vascular endothelial growth factor), all known to be regulated by the NF-kappaB. We found that anacardic acid inhibited both inducible and constitutive NF-kappaB activation; suppressed the activation of IkappaBalpha kinase that led to abrogation of phosphorylation and degradation of IkappaBalpha; inhibited acetylation and nuclear translocation of p65; and suppressed NF-kappaB-dependent reporter gene expression. Down-regulation of the p300 histone acetyltransferase gene by RNA interference abrogated the effect of anacardic acid on NF-kappaB suppression, suggesting the critical role of this enzyme. Overall, our results demonstrate a novel role for anacardic acid in potentially preventing or treating cancer through modulation of NF-kappaB signaling pathway.
Eukaryotic cell 2008 JUL
Histone acetyltransferase inhibitor anacardic acid causes changes in global gene expression during in vitro Plasmodium falciparum development.
Abstract
Abstract
To better understand the role of histone lysine acetylation in transcription in Plasmodium falciparum, we sought to attenuate histone acetyltransferase (HAT) activity using anacardic acid (AA). We showed that AA reversibly and noncompetitively inhibited the HAT activity of recombinant PfGCN5. To a lesser extent, AA inhibited the PfGCN5 activity in parasite nuclear extracts but did not affect histone deacetylase activity. AA blocked the growth of both chloroquine-sensitive and -resistant strains, with a 50% inhibitory concentration of approximately 30 microM. Treatment of the parasites with 20 microM of AA for 12 h had no obvious effect on parasite growth or gross morphology but induced hypoacetylation of histone H3 at K9 and K14, but not H4 at K5, K8, K12, and K16, suggesting inhibition of the PfGCN5 HAT. Microarray analysis showed that this AA treatment resulted in twofold or greater change in the expression of 271 (approximately 5%) parasite genes in late trophozoites, among which 207 genes were downregulated. Cluster analysis of gene expression indicated that AA mostly downregulated active genes, and this gene pool significantly overlapped with that enriched for H3K9 acetylation. We further demonstrated by chromatin immunoprecipitation and real-time PCR that AA treatment reduced acetylation near the putative promoters of a set of downregulated genes. This study suggests that the parasiticidal effect of AA is at least partially associated with its inhibition of PfGCN5 HAT, resulting in the disturbance of the transcription program in the parasites.
Journal of medicinal chemistry 2008 FEB
Specific small-molecule activator of Aurora kinase A induces autophosphorylation in a cell-free system.
Abstract
Abstract
Aurora kinases are essential for chromosomal segregation and cell division and thereby important for maintaining the proper genomic integrity. There are three classes of aurora kinases in humans: A, B, and C. Aurora kinase A is frequently overexpressed in various cancers. The link of the overexpression and tumorigenesis is yet to be understood. By employing virtual screening, we have found that anacardic acid, a pentadecane aliphatic chain containing hydroxylcarboxylic acid, from cashew nut shell liquid could be docked in Aurora kinases A and B. Remarkably, we found that anacardic acid could potently activate the Aurora kinase A mediated phosphorylation of histone H3, but at a similar concentration the activity of aurora kinase B remained unaffected in vitro. Mechanistically, anacardic acid induces the structural changes and also the autophosphorylation of the aurora kinase A to enhance the enzyme activity. This data thus indicate anacardic acid as the first small-molecule activator of Aurora kinase, which could be highly useful for probing the function of hyperactive (overexpressed) Aurora kinase A.

 网站首页
网站首页