概要
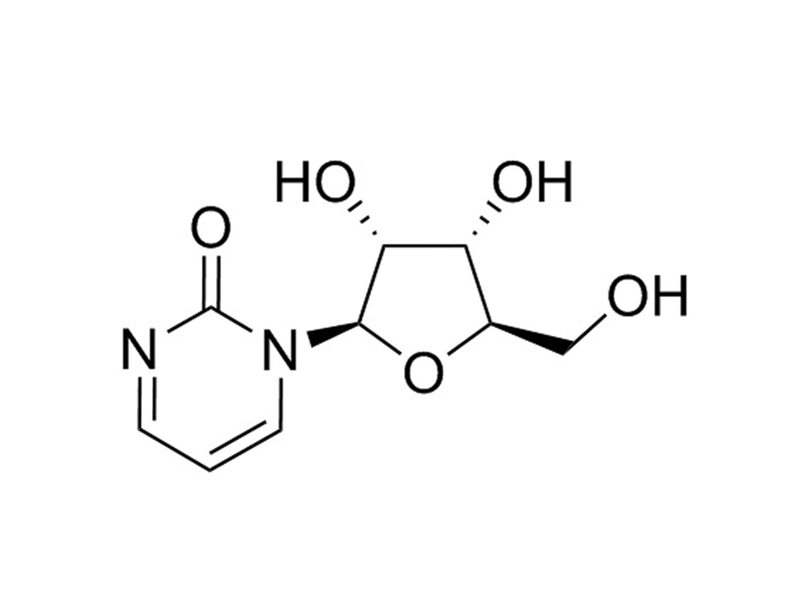
Zebularine, a cytidine analog, is a DNA methylation inhibitor that acts by forming a covalent complex with DNA methyltransferases (DNMTs; Zhou et al.).
DIFFERENTIATION
· Induces cardiomyocyte differentiation from rat bone marrow mesenchymal stem cells (MSCs; Naeem et al.).
CANCER RESEARCH
· Reduces proliferation of the cancer cell lines TK6, Jurkat, KG-1, and HCT116 (Stresemann et al.).
· Inhibits cell proliferation in T24 bladder carcinoma cells (Ben-Kasus et al.).
· In combination with the histone deacetylase inhibitor SAHA, reduces cell proliferation and increases apoptosis in pancreatic cancer cell lines (Neureiter et al.).
· Inhibits cell proliferation and induces apoptosis in human acute myeloid leukemia cells in vitro (Scott et al.).
· Increases the proportion of cells with cancer stem cell properties in subpopulations of human cancer cells and liver cancer cell lines (Marquardt et al.).
DIFFERENTIATION
· Induces cardiomyocyte differentiation from rat bone marrow mesenchymal stem cells (MSCs; Naeem et al.).
CANCER RESEARCH
· Reduces proliferation of the cancer cell lines TK6, Jurkat, KG-1, and HCT116 (Stresemann et al.).
· Inhibits cell proliferation in T24 bladder carcinoma cells (Ben-Kasus et al.).
· In combination with the histone deacetylase inhibitor SAHA, reduces cell proliferation and increases apoptosis in pancreatic cancer cell lines (Neureiter et al.).
· Inhibits cell proliferation and induces apoptosis in human acute myeloid leukemia cells in vitro (Scott et al.).
· Increases the proportion of cells with cancer stem cell properties in subpopulations of human cancer cells and liver cancer cell lines (Marquardt et al.).
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | Zebularine | 72902 | All | English |
| Safety Data Sheet | Zebularine | 72902 | All | English |
数据及文献
Publications (9)
Cardiovascular therapeutics 2013 AUG
DNA methylation inhibitors, 5-azacytidine and zebularine potentiate the transdifferentiation of rat bone marrow mesenchymal stem cells into cardiomyocytes.
Abstract
Abstract
BACKGROUND Mesenchymal stem cells (MSCs) have immense self-renewal capability. They can be differentiated into many cell types and therefore hold great potential in the field of regenerative medicine. MSCs can be converted into beating cardiomyocytes by treating them with DNA-demethylating agents. Some of these compounds are nucleoside analogs that are widely used for studying the role of DNA methylation in biological processes as well as for the clinical treatment of leukemia and other carcinomas. AIMS To achieve a better therapeutic option for cardiovascular regeneration, this study was carried out using MSCs treated with two synthetic compounds, zebularine and 5-azacytidine. It can be expected that treated MSCs prior to transplantation may increase the likelihood of successful regeneration of damaged myocardium. METHODS The optimized concentrations of these compounds were added separately into the culture medium and the treated cells were analyzed for the expression of cardiac-specific genes by RT-PCR and cardiac-specific proteins by immunocytochemistry and flow cytometry. Treated MSCs were cocultured with cardiomyocytes to see the fusion capability of these cells. RESULTS mRNA and protein expressions of GATA4, Nkx2.5, and cardiac troponin T were observed in the treated MSCs. Coculture studies of MSCs and cardiomyocytes have shown improved fusion with zebularine-treated MSCs as compared to untreated and 5-azacytidine-treated MSCs. CONCLUSION The study is expected to put forth another valuable aspect of certain compounds, that is, induction of transdifferentiation of MSCs into cardiomyocytes. This would serve as a tool for modified cellular therapy and may increase the probability of better myocardial regeneration.
Hepatology (Baltimore, Md.) 2011 SEP
Human hepatic cancer stem cells are characterized by common stemness traits and diverse oncogenic pathways.
Abstract
Abstract
UNLABELLED Epigenetic mechanisms play critical roles in stem cell biology by maintaining pluripotency of stem cells and promoting differentiation of more mature derivatives. If similar mechanisms are relevant for the cancer stem cell (CSC) model, then epigenetic modulation might enrich the CSC population, thereby facilitating CSC isolation and rigorous evaluation. To test this hypothesis, primary human cancer cells and liver cancer cell lines were treated with zebularine (ZEB), a potent DNA methyltransferase-1 inhibitor, and putative CSCs were isolated using the side population (SP) approach. The CSC properties of ZEB-treated and untreated subpopulations were tested using standard in vitro and in vivo assays. Whole transcriptome profiling of isolated CSCs was performed to generate CSC signatures. Clinical relevance of the CSC signatures was evaluated in diverse primary human cancers. Epigenetic modulation increased frequency of cells with CSC properties in the SP fraction isolated from human cancer cells as judged by self-renewal, superior tumor-initiating capacity in serial transplantations, and direct cell tracking experiments. Integrative transcriptome analysis revealed common traits enriched for stemness-associated genes, although each individual CSC gene expression signature exhibited activation of different oncogenic pathways (e.g., EGFR, SRC, and MYC). The common CSC signature was associated with malignant progression, which is enriched in poorly differentiated tumors, and was highly predictive of prognosis in liver and other cancers. CONCLUSION Epigenetic modulation may provide a tool for prospective isolation and in-depth analysis of CSC. The liver CSC gene signatures are defined by a pernicious interaction of unique oncogene-specific and common stemness traits. These data should facilitate the identifications of therapeutic tools targeting both unique and common features of CSCs.
PloS one 2010 JAN
Mechanistic insights on the inhibition of c5 DNA methyltransferases by zebularine.
Abstract
Abstract
In mammals DNA methylation occurs at position 5 of cytosine in a CpG context and regulates gene expression. It plays an important role in diseases and inhibitors of DNA methyltransferases (DNMTs)--the enzymes responsible for DNA methylation--are used in clinics for cancer therapy. The most potent inhibitors are 5-azacytidine and 5-azadeoxycytidine. Zebularine (1-(beta-D-ribofuranosyl)-2(1H)- pyrimidinone) is another cytidine analog described as a potent inhibitor that acts by forming a covalent complex with DNMT when incorporated into DNA. Here we bring additional experiments to explain its mechanism of action. First, we observe an increase in the DNA binding when zebularine is incorporated into the DNA, compared to deoxycytidine and 5-fluorodeoxycytidine, together with a strong decrease in the dissociation rate. Second, we show by denaturing gel analysis that the intermediate covalent complex between the enzyme and the DNA is reversible, differing thus from 5-fluorodeoxycytidine. Third, no methylation reaction occurs when zebularine is present in the DNA. We confirm that zebularine exerts its demethylation activity by stabilizing the binding of DNMTs to DNA, hindering the methylation and decreasing the dissociation, thereby trapping the enzyme and preventing turnover even at other sites.
Scandinavian journal of gastroenterology 2007 JAN
Apoptosis, proliferation and differentiation patterns are influenced by Zebularine and SAHA in pancreatic cancer models.
Abstract
Abstract
OBJECTIVE Pancreatic cancer continues to be an urgent clinical problem. We used the novel DNA methyltransferase inhibitor Zebularine and the histone deacetylase inhibitor SAHA to investigate the epigenetic influence on viability and differentiation of the pancreatic cancer cell lines YAP C, DAN G and Panc-89 in vitro and in vivo. MATERIAL AND METHODS Cell vitality, proliferation and expression of PDX-1, cytokeratin 7 and 20, chromogranin A, vimentin, bax and bcl-2 were determined on the protein and mRNA level in vitro and in a subcutaneous xenograft model. RESULTS A time- and dose-dependent increase of apoptosis, paralleled by decreased proliferation, was observed after incubation with single agents or a combination therapy with lower concentrations. This was associated with up-regulation of pro-apoptotic bax and a phenotypic stabilization by the enhanced expression of cytokeratin 7. In vivo, growth of xenografts was delayed with the most pronounced effect in Panc-89 after 1 week of daily intraperitoneal injections of Zebularine paralleled with CK7 up-regulation and down-regulation of dedifferentiation markers. CONCLUSIONS Epigenetic modulation via inhibition of DNA methyltransferase and histone deacetylase induces apoptosis in human pancreatic cancer cells in vitro and delays xenograft growth in vivo, which is associated with a morphological/molecular phenotypic stabilization. These compounds may therefore be suitable as adjunctive therapeutic agents in the treatment of pancreatic cancer.
Experimental hematology 2007 FEB
Zebularine inhibits human acute myeloid leukemia cell growth in vitro in association with p15INK4B demethylation and reexpression.
Abstract
Abstract
OBJECTIVE The p15INK4B tumor suppressor is frequently silenced by promoter hypermethylation in myelodysplastic syndrome and acute myeloid leukemia (AML). Clinically approved DNA methylation inhibitors, such as 5-aza-2'-deoxycytidine, can reverse p15INK4B promoter methylation, but widespread clinical use of these inhibitors is limited by their toxicity and instability in aqueous solution. The cytidine analog zebularine is a stable DNA methylation inhibitor that has minimal toxicity in vitro and in vivo. We evaluated zebularine effects on p15INK4B reactivation and cell growth in vitro to investigate a potential role for zebularine in treating myeloid malignancies. METHODS We examined the specific effects of zebularine on reexpression of transcriptionally silenced p15INK4B and its global effects on cell cycle and apoptosis in AML cell lines and primary patient samples. RESULTS Zebularine treatment of AML193, which has a densely methylated p15INK4B promoter, results in a dose-dependent increase in p15INK4B expression that correlates with CpG island promoter demethylation and enrichment of local histone acetylation. We observed enhanced p15INK4B induction following co-treatment with zebularine and the histone deacetylase inhibitor Trichostatin A. Zebularine inhibits cell proliferation, arrests cells at G(2)/M, and induces apoptosis at dosages that effectively demethylate the p15INK4B promoter. Zebularine treatment of KG-1 cells and AML patient blasts with hypermethylated p15INK4B promoters also reactivates p15INK4B reexpression and induces apoptosis. CONCLUSION Zebularine is an effective inhibitor of p15INK4B methylation and cell growth in human AML in vitro. Our results extend the spectrum of zebularine effects to nonepithelial malignancies and provide a strong rationale for evaluating its clinical utility in the treatment of myeloid malignancies.
Cancer research 2006 MAR
Functional diversity of DNA methyltransferase inhibitors in human cancer cell lines.
Abstract
Abstract
DNA methyltransferase inhibitors represent promising new drugs for cancer therapies. The first of these compounds (5-azacytidine, Vidaza) has recently been approved as an antitumor agent, and others are presently in various stages of their preclinical or clinical development. Most of the archetypal inhibitors have been established and characterized in different experimental systems, which has thus far precluded their direct comparison. We have now established defined experimental conditions that allowed a comparative analysis of the six most widely known DNA methyltransferase inhibitors: 5-azacytidine (5-aza-CR), 5-aza-2'-deoxycytidine (5-aza-CdR), zebularine, procaine, (-)-epigallocatechin-3-gallate (EGCG), and RG108. Of these, 5-aza-CR, 5-aza-CdR, zebularine, and EGCG were found to exhibit significant cytotoxicity in human cancer cell lines. 5-aza-CdR and EGCG were also found to be genotoxic, as evidenced by the induction of micronuclei. In addition, 5-aza-CR, 5-aza-CdR, zebularine, and RG108 caused concentration-dependent demethylation of genomic DNA, whereas procaine and EGCG failed to induce significant effects. Finally, the experiments in cancer cell lines were complemented by a cell-free in vitro assay with purified recombinant DNA methyltransferase, which indicated that RG108 is the only drug capable of direct enzyme inhibition. These results show a substantial diversity in the molecular activities of DNA methyltransferase inhibitors and provide valuable insights into the developmental potential of individual drugs.

 网站首页
网站首页