概要
The EasySep™ Mouse Plasmacytoid DC Isolation Kit is designed to isolate plasmacytoid dendritic cells (pDCs) from single-cell suspensions of splenocytes or other tissues by negative selection. Unwanted cells are targeted for removal with biotinylated antibodies that are directed against non-pDCs. Labeled cells are then recognized by Tetrameric Antibody Complexes that are directed against biotin and dextran. These cells are bound to magnetic particles and separated using an EasySep™ magnet without the use of columns. Desired cells are poured off into a new tube.
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | EasySep™ Mouse Plasmacytoid DC Isolation Kit | 19764 | All | English |
| Product Information Sheet | RoboSep™ Mouse Plasmacytoid DC Isolation Kit | 19764RF | All | English |
| Safety Data Sheet 1 | EasySep™ Mouse Plasmacytoid DC Isolation Kit | 19764 | All | English |
| Safety Data Sheet 2 | EasySep™ Mouse Plasmacytoid DC Isolation Kit | 19764 | All | English |
| Safety Data Sheet | RoboSep™ Mouse Plasmacytoid DC Isolation Kit | 19764RF | All | English |
数据及文献
Data
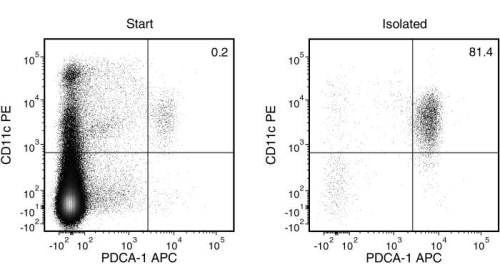
Figure 1. Typical EasySep™ Mouse Plasmacytoid DC Isolation Profile
Starting with mouse splenocytes, the pDC content (PDCA-1+CD11c+) of the isolated fraction typically ranges from 62 - 94%.
Publications (2)
Journal of immunology (Baltimore, Md. : 1950) 2016 JUL
FcγRIIB-Independent Mechanisms Controlling Membrane Localization of the Inhibitory Phosphatase SHIP in Human B Cells.
Abstract
Abstract
SHIP is an important regulator of immune cell signaling that functions to dephosphorylate the phosphoinositide phosphatidylinositol 3,4,5-trisphosphate at the plasma membrane and mediate protein-protein interactions. One established paradigm for SHIP activation involves its recruitment to the phospho-ITIM motif of the inhibitory receptor FcγRIIB. Although SHIP is essential for the inhibitory function of FcγRIIB, it also has critical modulating functions in signaling initiated from activating immunoreceptors such as B cell Ag receptor. In this study, we found that SHIP is indistinguishably recruited to the plasma membrane after BCR stimulation with or without FcγRIIB coligation in human cell lines and primary cells. Interestingly, fluorescence recovery after photobleaching analysis reveals differential mobility of SHIP-enhanced GFP depending on the mode of stimulation, suggesting that although BCR and FcγRIIB can both recruit SHIP, this occurs via distinct molecular complexes. Mutagenesis of a SHIP-enhanced GFP fusion protein reveals that the SHIP-Src homology 2 domain is essential in both cases whereas the C terminus is required for recruitment via BCR stimulation, but is less important with FcγRIIB coligation. Experiments with pharmacological inhibitors reveal that Syk activity is required for optimal stimulation-induced membrane localization of SHIP, whereas neither PI3K or Src kinase activity is essential. BCR-induced association of SHIP with binding partner Shc1 is dependent on Syk, as is tyrosine phosphorylation of both partners. Our results indicate that FcγRIIB is not uniquely able to promote membrane recruitment of SHIP, but rather modulates its function via formation of distinct signaling complexes. Membrane recruitment of SHIP via Syk-dependent mechanisms may be an important factor modulating immunoreceptor signaling.
Journal of immunology (Baltimore, Md. : 1950) 2013 MAR
Plasmacytoid dendritic cells play a key role in tumor progression in lipopolysaccharide-stimulated lung tumor-bearing mice.
Abstract
Abstract
The antitumor activity of LPS was first described by Dr. William Coley. However, its role in lung cancer remains unclear. The aim of our study was to elucidate the dose-dependent effects of LPS (0.1-10 μg/mouse) in a mouse model of B16-F10-induced metastatic lung cancer. Lung tumor growth increased at 3 and 7 d after the administration of low-dose LPS (0.1 μg/mouse) compared with control mice. This was associated with an influx of plasmacytoid dendritic cells (pDCs), regulatory T cells, myeloid-derived suppressor cells, and CD8(+) regulatory T cells. In contrast, high-dose LPS (10 μg/mouse) reduced lung tumor burden and was associated with a greater influx of pDCs, as well as a stronger Th1 and Th17 polarization. Depletion of pDCs during low-dose LPS administration resulted in a decreased lung tumor burden. Depletion of pDCs during high-dose LPS treatment resulted in an increased tumor burden. The dichotomy in LPS effects was due to the phenotype of pDCs, which were immunosuppressive after the low-dose LPS, and Th1- and T cytotoxic-polarizing cells after the high-dose LPS. Adoptive transfer of T cells into nude mice demonstrated that CD8(+) T cells were responsible for pDC recruitment following low-dose LPS administration, whereas CD4(+) T cells were required for pDC influx after the high-dose LPS. In conclusion, our data suggest differential effects of low-dose versus high-dose LPS on pDC phenotype and tumor progression or regression in the lungs of mice.

 网站首页
网站首页




