概要
cell-derived hematopoietic progenitor cells in defined serum-free conditions. MethoCult™ SF H4636 is formulated to support optimal growth of erythroid progenitor cells (CFU-E and BFU-E), granulocyte-macrophage progenitor cells (CFU-GM, CFU-G, CFU-M), and multipotential progenitor cells (CFU-GEMM; granulocyte, erythrocyte, macrophage, megakaryocyte).
MethoCult™ SF H4636 is also recommended for CFU assays with mononuclear cells, CD34+ enriched cells, and cells isolated by other purification methods from human bone marrow (BM), mobilized peripheral blood (MPB), peripheral blood (PB), and cord blood (CB) samples.
Browse our Frequently Asked Questions (FAQs) on performing the CFU assay and explore its utility as part of the cell therapy workflow.
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | MethoCult™ SF H4636 | 04636 | All | English |
| Safety Data Sheet | MethoCult™ SF H4636 | 04636 | All | English |
数据及文献
Data
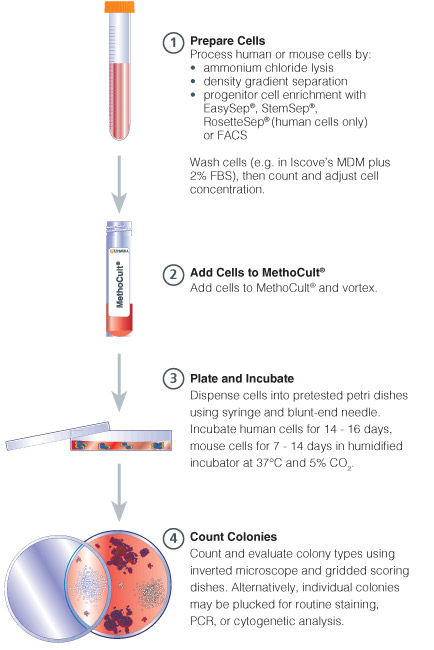
Figure 1. Procedure Summary for Hematopoietic CFU Assays
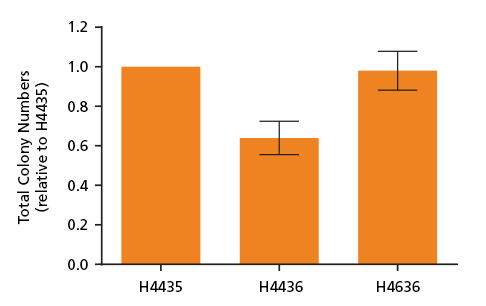
Figure 2. Total Colony Numbers of CD34+ Cells Grown in MethoCult™ SF H4436 and MethoCult™ SF H4636
CD34+ and mononuclear cells derived from CB, mPB and BM were isolated and plated in MethoCult™ media and counted after 14 days. Shown are the number of total colonies grown in serum-free MethoCult™ SF H4436 (Catalog #04436) and MethoCult™ SF H4636 (Catalog #04636) normalized relative to total colonies grown in serum-containing MethoCult™ H4435 (Catalog #04435). MethoCult™ SF H4636 provides improved conditions for colony growth in serum-free conditions as compared to MethoCult™ SF H4436. Shown are means with standard deviation (n = 6).

 网站首页
网站首页



